ISO 15189 2007 Medical laboratories Particular requirement
Home / Products/Services / Yellow Belt Certification Services / ISO 15189 2007 Medical laboratories Particular requirement
ISO 15189 2007 Medical laboratories Particular requirement
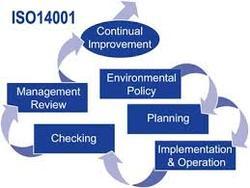
ISO 15189:2012 specifies requirements for quality and competence in medical laboratories. ISO 15189:2012 can be used by medical laboratories in developing their quality management systems and assessing their own competence. It can also be used for confirming or recognizing the competence of medical laboratories by laboratory customers, regulating authorities and accreditation bodies.ISO 15189 Medical laboratories — Particular requirements for quality and competence specifies the quality management system requirements particular to medical laboratories. The standard was developed by the International Organisation for Standardisations's Technical Committee 212 (ISO/TC 212). ISO/TC 212 assigned ISO 15189 to a working group to prepare the standard based on the details of ISO/IEC 17025:1999 General requirements for the competence of testing and calibration laboratories. This working group included provision of advice to users of the laboratory service, the collection of patient samples, the interpretation of test results, acceptable turnaround times, how testing is to be provided in a medical emergency and the lab\\\\\\\\\\\\'s role in the education and training of health care staff. While the standard is based on ISO/IEC 17025 and ISO 9001, it is a unique document that takes into consideration the specific requirements of the medical environment and the importance of the medical laboratory to patient care. Software solutions designed specifically for medical laboratories can aid in achieving ISO 15189 based accreditation. In particular, document control software can help by improving turnaround time (TAT) for document reviews, increasing efficiency of staff and improving overall quality.
Send EnquiryRelated Products
Products / Services
- Lead Auditor Training
- Lead Auditor Iso 22001 Fsms/haccp
- Gost R Explosion Proof Certificate
- Internal Auditor Training
- Testing Civil Construction Products
- Ce Marking Services
- Six Sigma Training Institute
- Six Sigma Executive Program
- Six Sigma Green Belt
- Six Sigma Black Belt
- La 9001training
- Six Sigma Training
- Training Program
- Welder Testing Certification
- Internal Auditor Iso 13485
- Internal Auditor Training
- Export To Uganda Pre-export Verification Of Conformity Std
- Internal Auditor Training
- Technical Inspection Tpi
- Security Inspection Machines
- Ship Technical Inspection Tpi
- Containers Technical Inspection Tpi
- Gostr Legal Framework
- Type Of Gost R Certificates
- What Is The Ukrsepro Certificate
- Type Of Ukrsepro Certificates
- Iso 9001 2008 Certification
- Information Technology Iso
- Iso 50001 Energy Management
- Iso 3834 Certification
- View All